Opened: 14 Nov 2008
Updated: 20 May 2010




Powered by boiling potassium! |
Opened: 14 Nov 2008 |
There are some other working fluids which have their own galleries in the Museum:
|
Potassium, a soft metal with a well-deserved reputation for exploding when dropped into water, is not the obvious choice for a working fluid. However, it is perfectly feasible to boil potassium and use the vapour to drive a turbine.
Like the mercury turbine, the motivation is to have a high heat-rejection temperature. This is essential in a space power-plant because heat can only be rejected by radiation, and a body needs to be hot to radiate effectively. Because of its high boiling point potassium can also be used to make a topping cycle for a steam plant in the same way as mercury.
Hot potassium is cerainly a hazardous substance, but it is not an insidious poison like mercury.
Potassium melts at 63.4 degC and boils at 759 degC; this is not ideal, as a working fluid that solidifies in the pipes when you switch the system off is somewhat less than convenient. This was underlined by the problems the Russians had with lead-bismuth cooled nuclear reactors in their Alfa-class submarines. A lead-bismuth eutectic solution solidifying at 125 °C (257 °F) was chosen as a higher coolant temperature gives an energy efficiency up to 1.5 times higher; it was also considered to be an advantage that the coolant would freeze if it leaked. However this caused major maintenance problems, and whenever a reactor was shut down, the liquid coolant had to be heated externally with superheated steam, or it would be impossible to restart the reactor, as the fuel assemblies would be frozen in the solid coolant. The first boat in service, K-64, appears to have suffered a frozen reactor at sea.
A good answer to the freezing problem is a mixture of sodium and potassium, (NaK) which has frequently been used as a coolant for nuclear reactors. When sodium/potassium alloys have between about 40% and 90% potassium by weight they are liquid at room temperature, which is a very handy thing in a cooling fluid. The mixture with the lowest melting point (the eutectic mix), consists of 78% potassium and 22% sodium, and this is liquid over the enormous range of -12.6 to 785 °C.
Remarkably, NaK technology has reached the desktop. A PC CPU cooler that uses a NaK liquid coolant has been released by www.danamics.com (2009) Not only that, but it circulates the metal with an electromagnetic pump having no moving parts, using a neodymium magnet. Cool! Reviews explicitly state that the eutectic NaK mixture is used.

POTASSIUM ENGINES
Considerable experience in potassium power-plants was built up by USA agencies, the aim being the generation of large amounts of power in space- far more than could be obtained from solar panel arrays, especially during missions at a long distance from the sun. Here is a fascinating and up-to-date (2004) document from The Oak Ridge National Laboratory. Note that on page 23 (document page numbers, not PDF numbers) two systems using mercury boilers (SNAP-2 and SNAP-8) are mentioned.
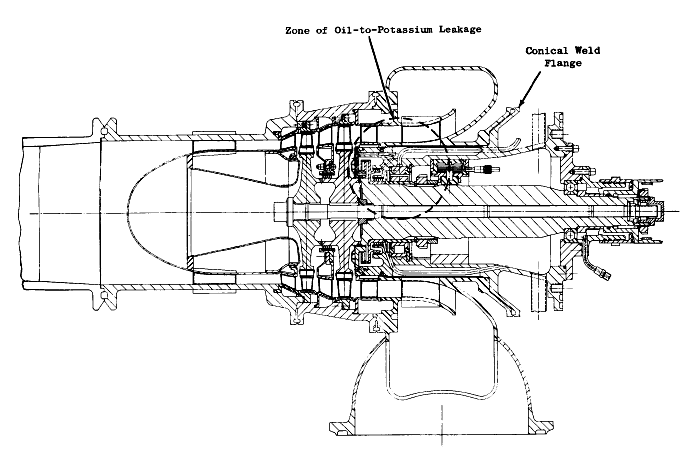 | Left: A two-stage potassium vapour turbine.
|

  
|